You are called urgently out of the ECG room. The receptionists have taken an elderly lady straight into a side room, when she was brought in to the hospital by her daughter.
You get as much history as possible from the daughter.
This is an 80 yr old lady who is new the clinic, and you have never met her before. The daughter explains she has become increasingly worried about her mother’s in recent months and has brought her to live nearer.
In the last few weeks she seems to have been getting generally weaker, and has been complaining of headaches and nausea. Her daughter has felt she was more low in mood, and more confused than usual. She was planning to book her an appointment to see you soon, but had been putting her decline down to her recent move, and was waiting to see if she picked up first.
On visiting her this morning, she found her quite ‘out-of-sorts’ and almost ‘vacant’ at times.
On examination she looks quite unwell. A thin, frail lady, who appears to be confused and has started vomiting.
She denies any chest pain.
You make a quick assessment and call the on call cardiac registrar.
Meanwhile cardiovascular and abdominal examination are unremarkable other than a bradycardia. Her BP is 100/60. She’s had no recent blood tests.
Her daughter tells you she has taken Atenolol ‘for many years’ for blood pressure. Other than that only some weekly ‘bone strengthening’ tablets and calcium/VitD supplements.
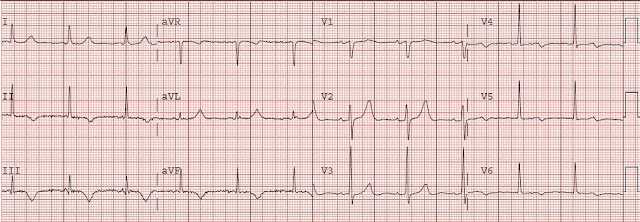
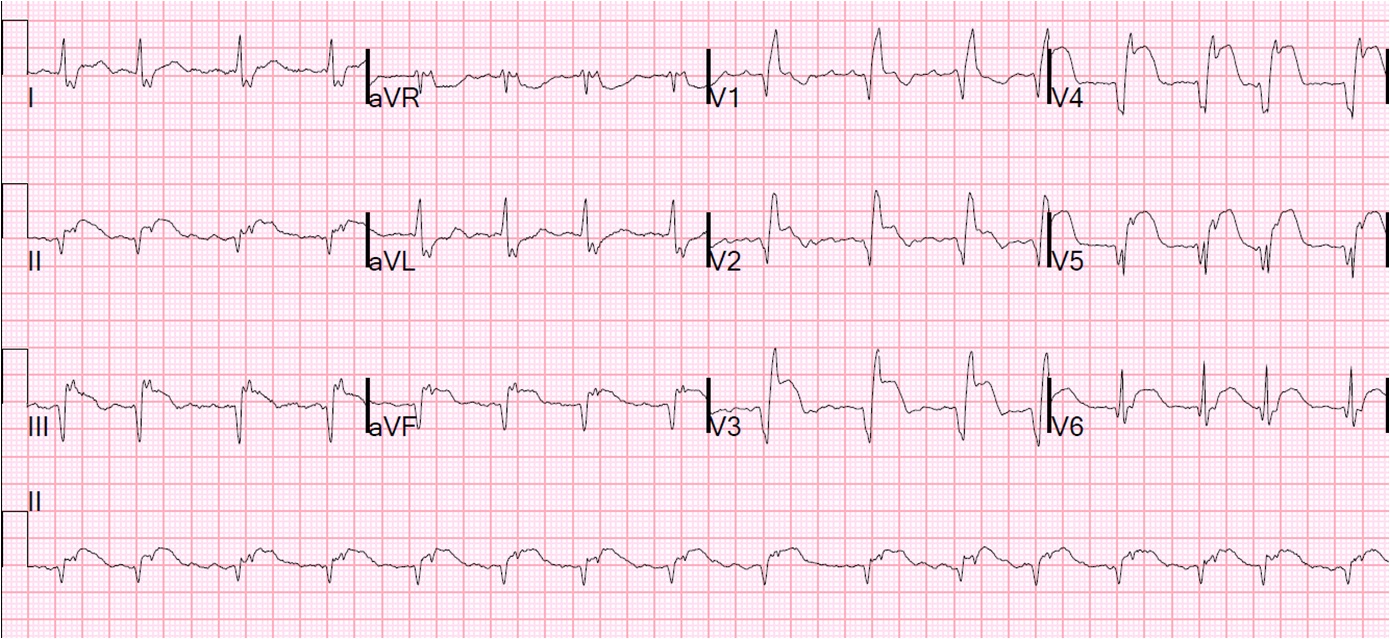
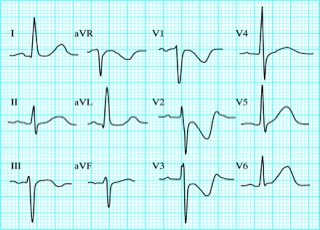
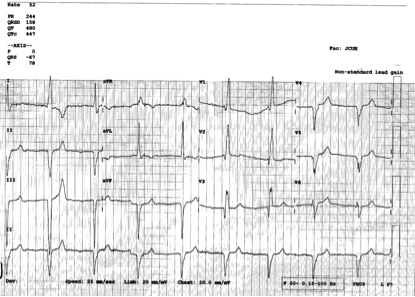
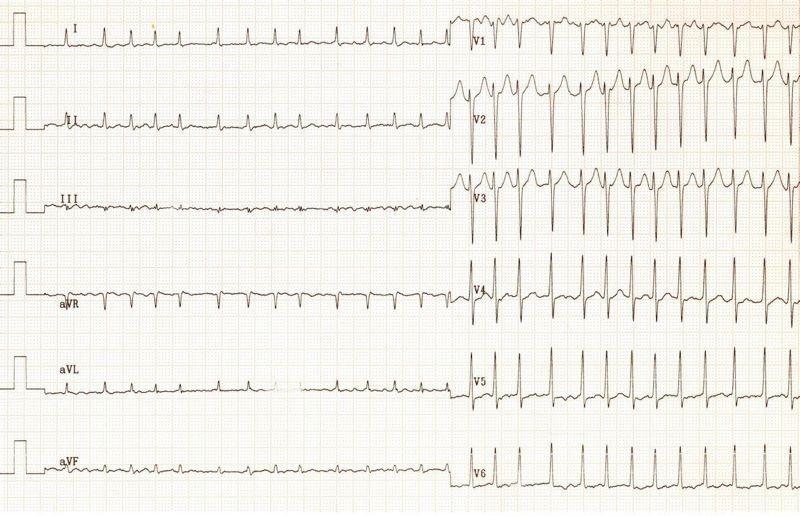
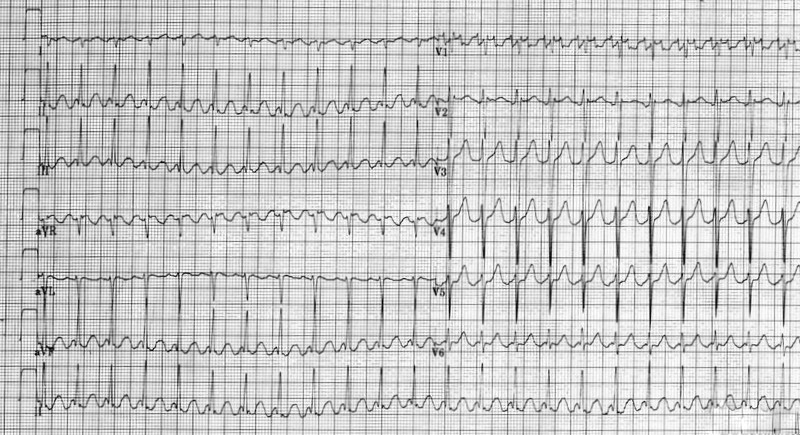
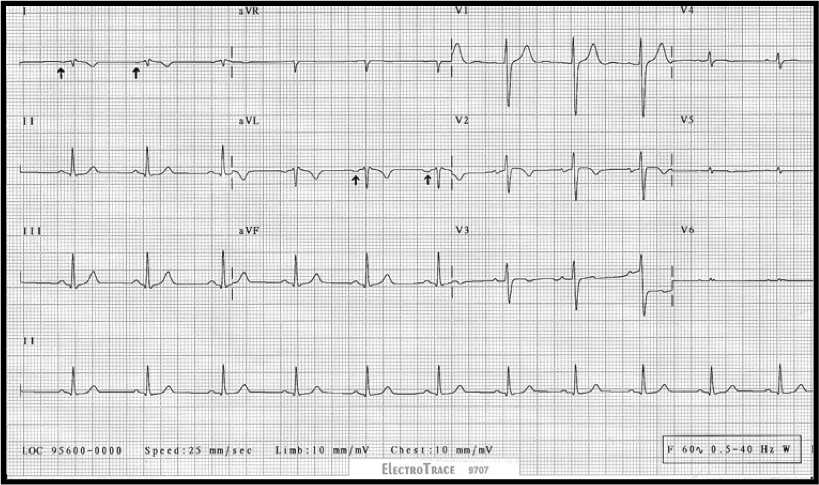
You do a 12 lead ECG:
What do you notice?
Q1. Can you see P waves? Is it regular? What is the rate?
What is the likely explanation?
Q2. Comment on the QRS complexes, ST segments and QT interval. (Hint: just describe what you see)
Discussion.
Q1. The heart rate is between 42-50 bpm (calculated crudely, by counting squares). This is of course assuming standard paper speed of 25mm/sec (not stated).
There are between 6 and 7 large squares between each QRS complex. So the rate is between 300÷6 and 300÷7
Using a rate-ruler, it’s 42 bpm 😉
So it’s a bradycardia.
I can’t make out any P waves, but the rhythm is regular.
This is most likley a junctional escape rhythm.
The Atenolol won’t be helping!
Q2. Firstly, the QRS complexes are a little wide (more than 1.5 small squares, but not > 3 small squares – as needed to define a bundle branch block). This suggests an intraventricular conduction defect. On it’s own, this can often be seen on ECGs, and may be of no signicicance.
More interesting, are the ST segments and QT interval.
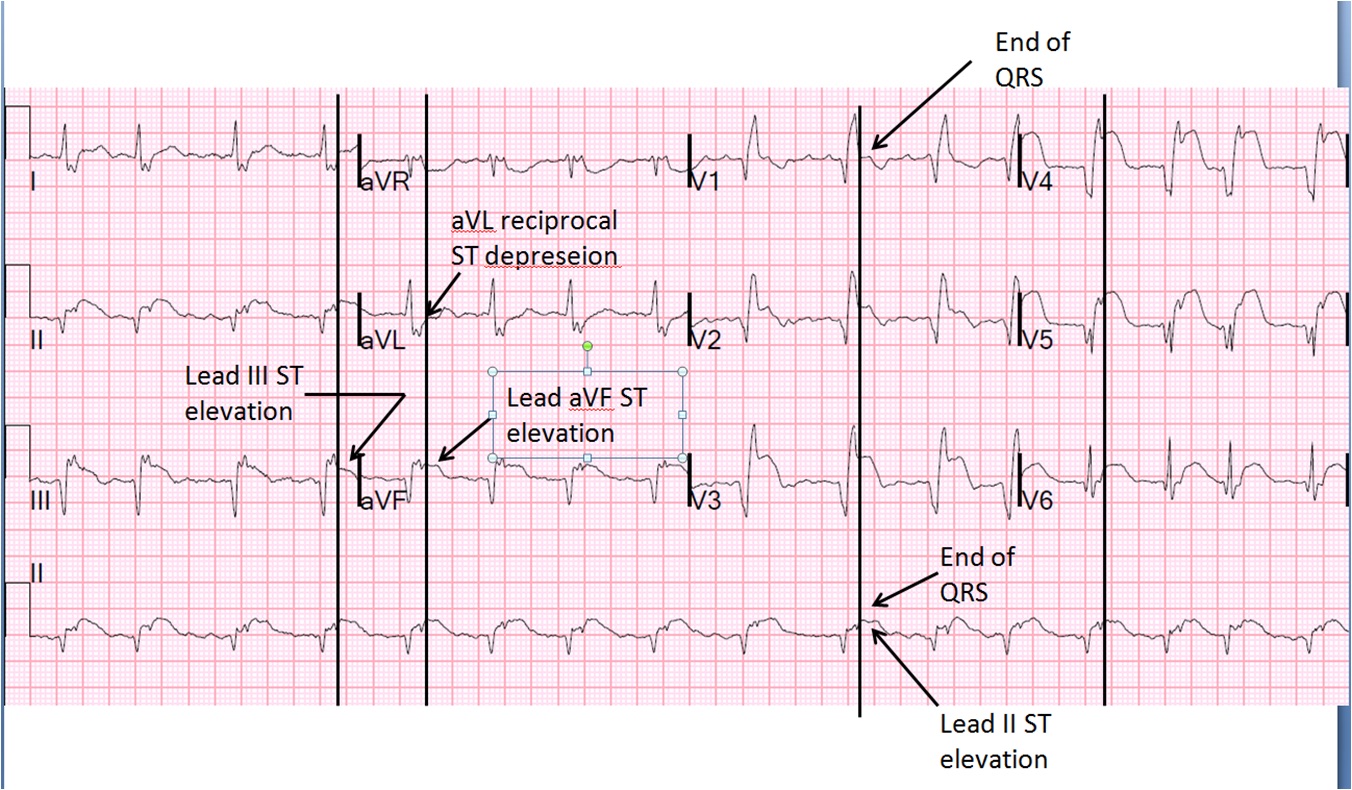
This ECG has a very short QT interval, with hardly any ST segment. This is the classic ECG picture of Hypercalcaemia.
Normal QT interval range: 0.36 -0.44 seconds (9-11 small squares)
On this ECG:
Corrected QT (QTC) = Bazett’s Formula = QT Interval / √ (RR interval).
Measured QT interval (ms) = 400
HR = 42
RR Interval (60 ÷ HR) = 1.4
√ RR = 1.2
QTC 334.7ms (0.33 sec)
HYPERCACAEMIA
Mild hypercalcaemia may have no symptoms or signs.
Symptoms of hypercalcaemia are more common at high serum calcium levels (12.0 mg/dL or 3 mmol/l). They include:
- Nausea & vomiting
- Alterations of mental status / lethargy / depression / headache/confusion
- Abdominal or flank pain
- Weakness and vague muscle/joint aches
- Polyuria, polydipsia, nocturia
Causes include
- Malignancy (via caused by increased osteoclastic activity within the bone)
- Primary hyperparathyroidism.
- Vit D toxicity (rare but recognised – see below)
Severe hypercalcaemia (above 15–16 mg/dL or 3.75–4 mmol/l) is considered a medical emergency, and cardiac arrest can result.
Electrolyte changes and the ECG can be remembered by recalling this simple sketch:
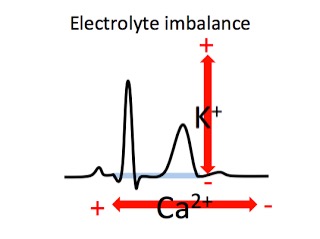
The T wave generally goes up and down with K+ level
The QT interval generally lengthens & shortens opposite to Ca2+ levels
Hypercalcaemia produces classical ECG changes of short ST segment, and a short QT interval. Sometimes the T wave is widened.
Hypocalcaemia gives the opposite effect, that is, a long ST segment and long QT interval.
Hyper- and Hypokalaemia – Generally, the amplitude of the T wave is proportional to the serum K+ level.
Is it possible, that this mildly confused lady, has been taking accidental overdoses of her calcium supplements, or even taking over the counter cod-liver oil in addition to her prescribed medication……???
Vitamin D Toxicty causing Hypercalcaemia with ECG changes.
This Pubmed link from July 2013 highlights the rare, but potentially fatal complication of Vitamin D3 (cholecalciferol) toxicity.
Severe hypercalcemia can sometimes mimic an acute Myocardial Infarction on ECG. This earlier Pubmed article, of 2003, presented a case of Vitamin D intoxication which mimicked acute myocardial infarction.
Hypercalcaemia can also mimic hypothermia, giving an ECG appearance of the Osborne wave.
Food for thought as more and more people become aware of the potential benefits of Vitamin D supplements.
See you next week.